Kuanming Yao1, Guangyao Zhao1,2, Xinge Yu1,3*
1 Department of Biomedical Engineering, City University of Hong Kong, Hong Kong SAR, People’s Republic of China
2 Institute of Materials Research, Tsinghua Shenzhen International Graduate School, Tsinghua University, Shenzhen 518055, China
3 Hong Kong Centre for Cerebro-Cardiovascular Health Engineering (COCHE), Hong Kong Science Park, Hong Kong SAR, People’s Republic of China
* Corresponding Author: xingeyu@cityu.edu.hk
Distinguished from conventional rigid electronics, soft electronics is becoming a novel platform for next-generation biomedical instrumentations. With advanced materials, mechanics, and structural design, soft electronics could be realized in thin, light-weighted formats and thus can be worn on or implanted in human body, and may excel in great stretchability and conformal attachment with skin or tissue, which ensures continuous and precise healthcare monitoring or therapies. Our group focuses on exploring the novel soft electronics for the applications in various fields of biomedical applications, including motion and mechanical sensing, wearable energy harvesting, dynamic temperature sensing, sweat sensing, and closed-loop human-machine interface (Fig.1.a).
Recent advances in biomedical applications of soft electronics.
One of the basic functions of wearable soft electronics is sensing mechanical information. Pressure, strain, motions, and mechano-acoustic information of humans can be acquired by piezoresistive, pizeocapactive, piezoelectric, or triboelectric mechanisms. Commercial mechanical sensing solutions such as tensile testers and ultrasonic probes were usually bulky with complicated structures, which makes them inconvenient for use in daily life, thus requires a flexible and light-weighted substitute. To realize the motion and tactile sensing on the body, our group reported a high-throughput and scalable electronic skin based on stretchable piezoelectric lead zirconate titanate (PZT) elastomer by screen printing fabrication. This electronic skin can be integrated on body for motion recognition, and enables the real-time replication of gestures with a robotic hand by sensing the joint strains and encodes into corresponding motion commands. This e-skin in dense sensing array style can acquire delicate tactile information, such as finger touching, with high spatial resolution (25 devices /cm-2) and superior sensitivity (180µV/kPa) (Fig.1.b left)1. Driven by the practical application requirements, our group developed a miniaturized electromechanical system for quick characterization of the biomechanics of skin and deep tissues by combining strain sensors and actuators in one piece. Benefiting from the skin deformation caused by vibration of the actuators, fluctuation of sensor resistance at the same frequency could be detected to characterize the strain, and thus can obtain mechanical properties of millimeter-thick tissues under the skin (Fig.1.b right), which helps quick diagnosis of skin diseases like psoriasis and skin cancer, and has a promising prospect in kinesiology and rehabilitation2.
These sensed mechanical and other kinds of energy could also be harvested for usage, since that in situ powering systems are critical for wearable electronics or implantable devices, which consist of a series of energy-consuming components. These powering systems need to be thin, soft, and energy dense for better integration with soft electronic devices. To solve this problem, our group developed some ultra-thin and stretchable self-powered sensors capable of energy harvesting and health monitoring, which achieved natural and imperceptible integration with skin. These sensors are based on various mechanisms such as biofuel cells, piezoelectric, and triboelectric nanogenerators. For instance, one of our recent work reported an enzymatic epidermal self-powered sweat sensor. With the effective sweat collection through microfluidic channels, the device interacting with sweat served both as a biofuel cell and the accurate sensing component for in situ detection of lactate and glucose, which avoided using additional power supplies3 (Fig.1.c left). Exploring new materials4 and designing device structures5-10 can also efficiently convert mechanical energy into electricity. Among the current energy harvesting materials/devices, triboelectric nanogenerators (TENG) have been proven to be an excellent candidate for energy generating through straightforward contact-electrification and electrostatic induction effect, yet they are usually too thick to be integrated on skin unobtrusively. To solve this problem, we developed an ultra-thin, highly flexible, aesthetic, and stretchable, tattoo-like triboelectric nanogenerator (TL-TENG) by ultra-thin liquid bandage and delicate pattern design, which can be integrated intimately and imperceptibly with the human skin6 (Fig.1.c right). The TL-TENG collects mechanical energy from body activities and transforms it into electricity. Therefore, the platform can also be designed as self-powering sensors for human-machine interfaces, such as wirelessly controlling robots, which demonstrates great potential in powering wearable smart systems and the internet of things.
Dynamic body thermal monitoring is also a very important strategy for the real-time sensing of human health statuses, such as human respiration and hemodynamic monitoring. Recently, we demonstrated a highly flexible, stretchable, and lightweight thermosensitive respiration sensor, with a two-dimensional stretchable fractal curve design, sensitive thermistor, and active sensing strategy11 (Fig.1.d left). The device can conformably integrate with skin even under large mechanical deformations and detect the respiration rate/depth during physical exercise, rests, and prolonged rest (siesta), which indicates the feasibility in future diagnostic breath analysis and respiration status recognition. Besides, for detecting the subcutaneous hymodynamic status without large medical imaging equipment which are bulky, expensive and inconvenient, we also developed an ultra-thin, noninvasive, and flexible thermal-sensing electronic skin (e-skin) to monitor the thermal properties of blood vessels by a dense array of thermal sensors12 (Fig.1.d right). During the on-body experiment, the bare epidermis and the blood vessel could be exactly distinguished and even the flow direction could be detected at the same time. Enpowered by 4 multiplexers and BLE communication for wireless high-throughput sensing, it independently senses vascular hemodynamics without imaging equipment, which has great utility as a portable diagnostics device for point-of-care.
Sweat contains lots of physiological biomarkers, thus continuous bio-sensing of sweat could help monitor the health conditions during exercises. However, sweat analyzing techniques usually need equipment with power cords connected, which is inconvenient for sports scenarios. In situ power source that activated by sweat could help realize portable and wearable sweat sensing platforms. We developed a series of novel sweat-activated batteries (SABs) associated with functional electronics for smart sensing of sweat and other physiological indicators. For example, a stretchable Zn/Cu SAB cells array could be integrated on athletes’ skin and absorb sweat to trigger redox reactions, which provide power for 6- hour continuous sensing of glucose, Na+, and pH value via Bluetooth-embedded microelectronics13 (Fig.1.e left). It could also light up 120 LEDs for over 5 hours which can be used as a passive safety warning device for night running. To shrink the size of the device for better fitting with skin, we built a thin Mg/O2 SAB in a breathable tape that can fit into sportswear14 (Fig.1.e mid). It utilized a KCl-containing cotton sheet as rapid sweat absorber, electrolyte and salt bridge simultaneously. It’s thin and robust to stably work even during repeatedly severe bending, which enables an NFC-embedded sensor tag to monitor sweat Na+/pH and the skin hydration level at the joints by measuring skin impedance. To maximize the catalytic efficiency in oxygen reduction, we further used Ni foam coated with graphene for providing enhanced air diffusing performance in Mg/O2 SAB15 (Fig.1.e right). It has a power density that reaches 16.3 mW·cm-2 and an impressive energy capacity of 74.4 mAh, which allows supporting microelectronics for sensing of oxygen saturation in blood, pulse rate, body surface temperature, and exercising intensity over hours.
With the development of various sensors and power sources, we also made some efforts in the system-level integration work for biomedical applications, especially constructing a closed-loop of sensing, controlling, and perceiving. The outbreak of COVID-19 pandemic makes it necessary to reduce intimate social contact among people. An efficient human-machine interface (HMI) that helps people feel things virtually or remotely could help a lot in this context. We developed a skin-integrated wireless haptic interface that provides tactile feedback to the skin by an array of electromagnetic actuators16. In form of a “second skin”, it helps users perceive virtual haptic information in scenarios of VR/AR, gaming, and video calls, or helps amputees feel real objects through prosthetics (Fig.1.f left). The actuator delivers vibration to the skin based on Lorentz force by the interaction between a magnet and the alternating magnetic field generated by a coil. It can efficiently stimulate mechanoreceptors underlying the skin by maximizing vibration amplitude in optimized frequency. While dimensions of the actuator are still too large for reproducing feedback in fine resolution, its size could be shrunk to millimeter scale to fit an array into a soft band that could wrap on a fingertip to provide programmable and recognizable braille feedback for the blinds17 (Fig.1.f mid). Combining this feedback technology with motion sensing devices, we carried out an e-skin-based closed-loop HMI, where the user could both remotely control movement of robotics and perceive what robotics touch simultaneously through Bluetooth, Wi-Fi or Internet18 (Fig.1.f right). It helps commit precise medical tasks remotely through robotics, like collecting saliva samples for COVID-19 detection, nursing patients in non-contact ways, etc.
Conclusions and outlooks
In summary, we made efforts in developing soft electronics in the forms of wearable devices, electronic skins, and energy generators for various biomedical applications, from body fluid analysis, skin disorder diagnosis, to sensory recovery assisting, and boosting remote healthcare services in the future. We believe that through dedicated interdisciplinary teamwork, including medical, materials science, mechanical, electrical, and computational engineering fields, there are infinite possibilities for what soft electronics can do for medical purposes in the future.
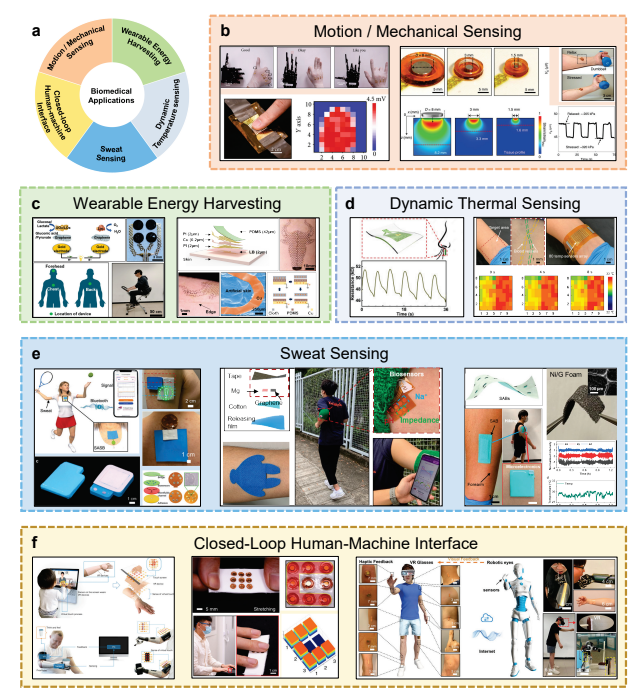
Figure 1. Examples of novel soft electronics for biomedical applications.
References
- Liu, Y, et al., Electronic Skin from High-Throughput Fabrication of Intrinsically Stretchable Lead Zirconate Titanate Elastomer. Research 2020; 2020: 1085417. DOI:10.34133/2020/1085417.
- Song, E, et al., Miniaturized electromechanical devices for the characterization of the biomechanics of deep tissue. Nat Biomed Eng 2021; 5 (7): 759-771. DOI:10.1038/s41551-021-00723-y.
- Huang, X, et al., Epidermal self-powered sweat sensors for glucose and lactate monitoring. Bio-Design and Manufacturing 2021; 5 (1): 201-209. DOI:10.1007/s42242-021-00156-1.
- Liu, Y, et al., Skin‐Integrated Graphene‐Embedded Lead Zirconate Titanate Rubber for Energy Harvesting and Mechanical Sensing. Advanced Materials Technologies 2019; 4 (12): 1900744. DOI:10.1002/admt.201900744.
- Yao, K-M, et al., Stretchable self-powered epidermal electronics from piezoelectric rubber for tactile sensing. Acta Physica Sinica 2020; 69 (17): 178701. DOI:10.7498/aps.69.20200664.
- Wong, T H, et al., Triboelectric Nanogenerator Tattoos Enabled by Epidermal Electronic Technologies. Advanced Functional Materials 2021: 2111269. DOI:10.1002/adfm.202111269.
- Wu, M, et al., Thin, soft, skin-integrated foam-based triboelectric nanogenerators for tactile sensing and energy harvesting. Materials Today Energy 2021; 20: 100657. DOI:10.1016/j.mtener.2021.100657.
- Li, J, et al., Thin, soft, 3D printing enabled crosstalk minimized triboelectric nanogenerator arrays for tactile sensing. Fundamental Research 2022. DOI:10.1016/j.fmre.2022.01.021.
- He, J, et al., Trampoline inspired stretchable triboelectric nanogenerators as tactile sensors for epidermal electronics. Nano Energy 2021; 81: 105590. DOI:10.1016/j.nanoen.2020.105590.
- Yao, K, et al., Mechanics designs-performance relationships in epidermal triboelectric nanogenerators. Nano Energy 2020; 76: 105017. DOI:10.1016/j.nanoen.2020.105017.
- Liu, Y, et al., Epidermal electronics for respiration monitoring via thermo-sensitive measuring. Materials Today Physics 2020; 13: 100199. DOI:10.1016/j.mtphys.2020.100199.
- Park, W, et al., High Channel Temperature Mapping Electronics in a Thin, Soft, Wireless Format for Non-Invasive Body Thermal Analysis. Biosensors 2021; 11 (435): 1-14. DOI:10.3390/bios11110435.
- Liu, Y, et al., Stretchable Sweat-Activated Battery in Skin-Integrated Electronics for Continuous Wireless Sweat Monitoring. Adv Sci 2022: e2104635. DOI:10.1002/advs.202104635.
- Huang, X, et al., Garment embedded sweat-activated batteries in wearable electronics for continuous sweat monitoring. npj Flexible Electronics 2022; 6 (1): 1-8. DOI:10.1038/s41528-022-00144-0.
- Liu, Y, et al., Bandage based energy generators activated by sweat in wireless skin electronics for continuous physiological monitoring. Nano Energy 2022; 92: 106755. DOI:10.1016/j.nanoen.2021.106755.
- Yu, X, et al., Skin-integrated wireless haptic interfaces for virtual and augmented reality. Nature 2019; 575 (7783): 473-479. DOI:10.1038/s41586-019-1687-0.
- Li, D, et al., Miniaturization of mechanical actuators in skin-integrated electronics for haptic interfaces. Microsyst Nanoeng 2021; 7 (85): 1-9. DOI:10.1038/s41378-021-00301-x.
- Liu, Y M, et al., Electronic skin as wireless human-machine interfaces for robotic VR. Sci Adv 2022; 8 (2): eabl6700. DOI:10.1126/sciadv.abl6700.


