RESEARCH
October 2019
Josef Faller, Jennifer Cummings, Sameer Saproo, Paul Sajda
High arousal can adversely affect task performance. Walking over a balance beam that sits 10 cm over the floor, for example, will be easier for most people than walking over a beam that is fixed at a height of 10 m, where a misstep could lead to grave injury. Another example is referred to as “pilot induced oscillations” (PIOs), where airplane pilots – under high arousal – dangerously overcompensate for small control errors in a way that can quickly escalate to losing control over and/or crashing the plane. In 1908, Yerkes & Dodson first formally described an inverse U-shape relationship between arousal and performance under high task difficulty [1]( see Figure 1.A). From the perspective of neurophysiology, there is evidence in support of the hypothesis that an interplay between the anterior cingulate cortex (ACC) and locus coeruleus (LC) – regions implicated in monitoring task performance and mediating stress responses – may play a critical role in explaining this phenomenon [2,3](see Figure 1.B). In a previous study, our group identified EEG signatures of PIO propensity or task-dependent arousal in a virtual flight task, a so called “boundary avoidance task” (BAT), where difficulty progressively increases over 90 seconds to induce PIOs and task failure, i.e. crashing the plane into a boundary [4](see Figure 1.C).
We asked whether we could decode task-dependent arousal in the same BAT paradigm from EEG in real-time, such that the output could be fed back to the subject in a closed-loop setup [5]. The idea was that subjects could potentially use the feedback in the form of an audio signal to down-regulate their own arousal in real-time, thus hypothetically mitigating the detrimental effects of high arousal such that task performance would increase relative to silence or sham feedback (see Figure 1. A, B & C).
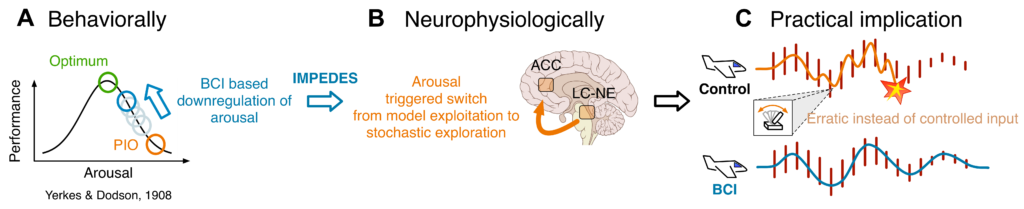
Figure 1: (A) Assuming that the performance decrease, when arousal levels spike during pilot induced oscillations (PIO), is goverend by the Yerkes & Dodson law, then brain-computer interface (BCI) based down-regulation of arousal should increase performance. (B) From the perspective of neurophysiology, down-regulating arousal is assumed to postpone a locus coeruleus (LC) triggered switch from model exploitation to stochastic exploration. (C) Hypothetically, down-regulation of arousal should lead to lower PIO propensity and thus higher task performance. Figure from [5].
To test this hypothesis we enrolled 20 subjects in an EEG experiment, where they would first attempt flights in the BAT paradigm for a total of 10 minutes. The EEG data collected during these flight attempts was subsequently used to train a linear, subject specific decoder that would output an index of inferred task-dependent arousal (see Figure 2.B). In the main experimental block, subjects alternately attempted flights in BAT courses of two difficulty levels (“easy” and “hard”), while the experimental condition for every flight attempt was drawn randomly from a uniform distribution over the conditions “BCI”, “Sham” and “Silence”. In condition BCI, the inferred level of task-dependent arousal from the EEG-based decoder modulated the loudness of audio of a constant-rate heart-beat. Louder heart-beat audio corresponded to a higher inferred level of task dependent arousal or inferred PIO propensity, while the audio became almost inaudible for low levels of inferred task dependent arousal. The sham audio was composed in equal parts of veridical decoder output and a random signal with matching spectral characteristics.
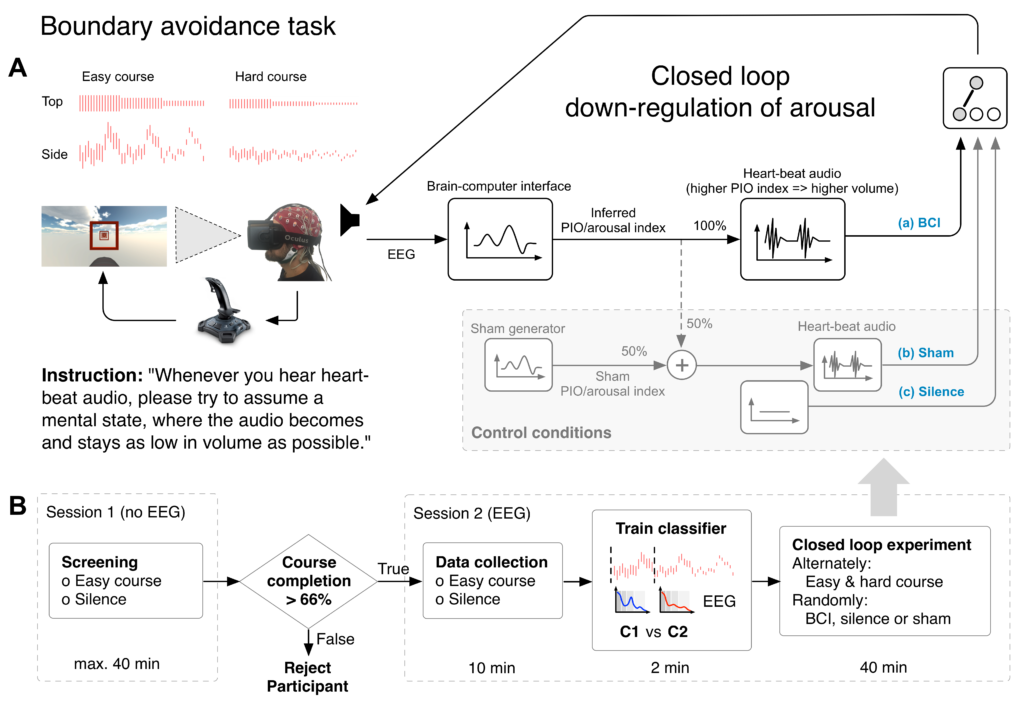
Figure 2: (A) Participants alternately navigated a virtual plane through an easier or more difficult course. One flight attempt was a maximum of 90 s long but participants typically failed much earlier since task difficulty increased every 30 s by means of a reduction in boundary size. One of the three experimental conditions “BCI”, “Sham” or “Silence” was randomly selected for every flight attempt. For BCI, inferred task dependent arousal modulated loudness of an audio signal such that louder audio corresponded to higher arousal. Sham feedback was generated in equal parts from veridical and random signal, while no audio was presented during Silence. (B) Participants who passed screening were admitted to the main experimental session, where the linear decoder was trained on 10 min of data based on spectral features. Figure from [5].
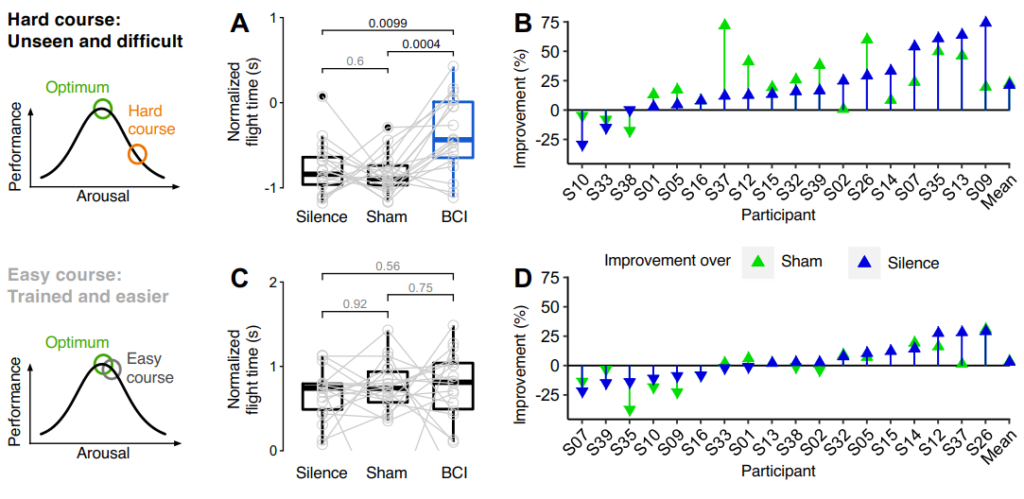
Figure 3: On the left we show the hypothesized operation points in terms of arousal and performance for the two course difficulties as per the law by Yerkes & Dodson. (A) Flight length was significantly longer with veridical feedback relative to control conditions and (B) shows that the effect is present across subjects. In accordance with the law by Yerkes & Dodson, no systematic performance difference between conditions was observed for the easy course in (C) or (D). The numbers over the brackets represent uncorrected p-values from t-tests for dependent samples. Figure from [5].
Acknowledgements
We thank Yida Lin for help with figures and formatting and James McIntosh for helpful discussion. This work was supported by Defense Advanced Research Projects Agency and Army Research Office Grant W911NF-16-1-0507, Army Research Laboratory Cooperative Agreement W911NF-10-2-0022, National Science Foundation Grant IIS-1527747, and Economic and Social Research Council Grant ES/ L012995/1. The views and conclusions contained in this document are those of the authors and should not be interpreted as representing the official policies, either expressed or implied, of the US Government. The US Government is authorized to reproduce and distribute reprints for Government purposes notwithstanding any copyright notation herein.
References
[1] Yerkes RM, Dodson JD (1908) The relation of strength of stimulus to rapidity of habit- formation. J Comp Neurol Psychol 18:459–482. [2] Aston-Jones G, Cohen JD (2005) An integrative theory of locus coeruleus-norepinephrine function: Adaptive gain and optimal performance. Annu Rev Neurosci 28:403–450. [3] Tervo DGR, et al. (2014) Behavioral variability through stochastic choice and its gating by anterior cingulate cortex. Cell 159:21–32. [4] Saproo S, Shih V, Jangraw DC, Sajda P (2016) Neural mechanisms underlying cata- strophic failure in human-machine interaction during aerial navigation. J Neural Eng 13:066005. [5] Faller, J, Cummings, J, Saproo, S, Sajda, P (2019) Regulation of arousal via online neurofeedback improves human performance in a demanding sensory-motor task. Proceedings of the National Academy of Sciences 116:6482-6490. [6] Gilzenrat MS, Nieuwenhuis S, Jepma M, Cohen JD (2010) Pupil diameter tracks changes in control state predicted by the adaptive gain theory of locus coeruleus function. Cogn Affect Behav Neurosci 10:252–269. [7] Joshi S, Li Y, Kalwani RM, Gold JI (2016) Relationships between pupil diameter and neu- ronal activity in the locus coeruleus, colliculi, and cingulate cortex. Neuron 89:221–234. [8] Berntson GG, Cacioppo JT (2004) Heart rate variability: Stress and psychiatric conditions. Dynamic Electrocardiography, eds Malik M, Camm AJ (Wiley, New York), pp 57–64. [9] Keynan JN, et al. (2016) Limbic activity modulation guided by functional magnetic resonance imaging-inspired electroencephalography improves implicit emotion regulation. Biol Psychiatry 80:490–496.


