RESEARCH
Dr. Salvatore Domenic Morgera
The human nervous system provides energy efficient, highly complex realization and control of how we sense, think and act. For machines designed by humans, the ideas of energy efficiency and complexity are at odds, thus the question of how the central nervous system (CNS) really works has received intense scrutiny for decades. Researchers at the University of South Florida (USF) under the direction of Dr. Sal Morgera have discovered a sophisticated electric near-field generated in an energy efficient, natural manner by our billions of myelinated nerve fibers. This electric near-field is roughly the counterpart of the magnetic near-field used in smartphone contactless payment services such as Apple Pay® and Google Wallet®, known as Near Field Communications, or NFC.
This work is an entirely new perspective on CNS structure and function. The researchers have discovered, for example, that the optic nerve has over one million electric near-field circuits forming a neurological network of extraordinary sophistication in which individual nerve fibers not only propagate action potentials as we learned in our high school biology texts, but also interact through electric near-fields1-3. This paradigm shift in understanding can be a game changer in the development, not only of artificial eyes and prosthetic limbs, but also of less invasive, clinically accurate neurodevices. Devices that passively read and actively alter the electric field structures of regions of the CNS and provide therapeutic treatment for the millions that suffer from neurological dysfunction and mental health disorders.
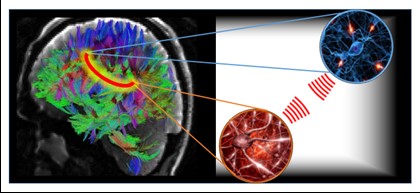
The near-field network is illustrated in the figure. On the left-half is the human connectome (http://www.humanconnectomeproject.org/), shown as a tractographic map of nerve fiber connections in the human brain. This mapping of anatomical connections is the fixed network of the brain. It is well known that this fixed brain network is not entirely successful in explaining brain function, especially for high-level cognitive tasks1,4,5. On the right-half of the graphic are blowups showing neurons in two representative regions of the brain. The discovery is the presence of a biologically significant electric near-field between these neurons, or neuronal groups, a point illustrated by the red spherical waves. These electric near-field waves communicate fine details of their respective regions in a bi-directional manner. This creates a wireless connection, signified by the red loop across these brain regions in the left-half. Thus, the brain consists of a fixed network augmented by a wireless network which helps to explain how physically disparate regions of the brain connect so quickly during the execution of certain cognitive tasks. Furthermore, neurological diseases, such as Multiple Sclerosis, Parkinson’s, and Autism Spectrum Disorder, have now been associated with dysfunctions that are like those experienced in conventional wireless networks.
This work was entered in the 2015 Alcatel-Lucent (now, Nokia) Bell Labs Prize competition. The competition attracted a wide range of industry impacting ideas from 33 countries that were whittled down to seven innovative ideas, one of which was this work on near-field coupling in the brain. Since that time, the ideas presented have been largely verified by in vitro experimentation, and the understanding of how the brain really works has been refined. The in vitro experimentation has been conducted both at low frequencies up to 100 kHz using AM Stimulus Isolation Units in a manner like6 and at high frequency at wavelengths of approximately 1600 nm using shortwave infrared equipment like that described in 7. The latter work is relevant, as it helps to predict photon penetration, essential to optical imaging and treatment using light-based therapies, as is the work presented in 8, where conductance can be viewed as a surrogate for connectivity.
The long-term plan is to passively read the signatures of the internal electric near-fields (the endogenous fields) using a custom designed antenna array to diagnose neurological dysfunction and then to rationally design time-varying, frequency-rich and spatially directed external electric fields (the exogenous fields) to actively interact with the internal fields and treat the dysfunction by “tuning” the appropriate region of the CNS. The idea of a rational design is one based on engineering and scientific facts2,6,9. This type of design is lacking in currently available neurological medical devices, and we believe is a deficiency that has led to significant numbers of recalls, lack of efficacy, and unexpected side effects.
Our work in this area was invited for a Keynote Presentation at the Nanotech France 2018 International Conference and Exhibition, June 27-29, 2018, held at the Pôle Universitaire Léonard de Vinci (www.devinci.fr/en) in Paris. Details were presented on the first step of the plan which is to characterize the brain’s electric near-fields more precisely. Both in silico computer models and in vitro measurement as described above suggest the following properties for the electric near-fields:
- The electric near-fields due to activity in myelinated nerve fibers appear to primarily occur in the wavelength range [300,2500] nm. This is the ultraviolet (4.1 eV) to infrared (0.5 eV) regime. They are essentially light-wave or photonic fields.
- The waves appear to be spherical (near-field), do not radiate freely, and are directed transversely into extracellular space at neighboring nerve fibers.
- Electric near-field levels at closely packed neighboring nerve fibers are as high as 10^4 V/m, a biologically significant level.
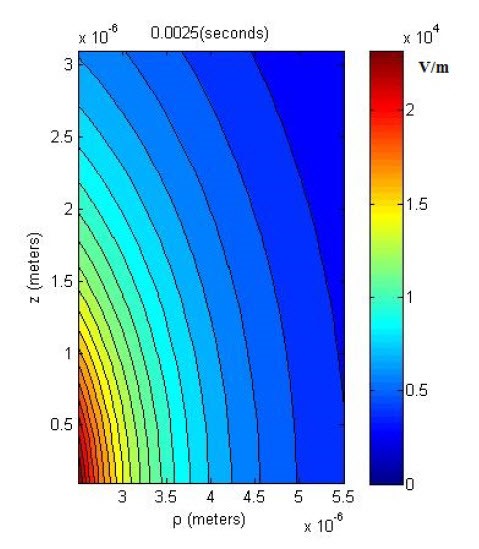
A graphic showing a measurement of the average electric near-field levels outside a node of Ranvier of an active, myelinated nerve fiber is shown below. We see that at 4 microns from the surface of the fiber the average electric near-field level is approximately 10^4 V/m (average cell phone level is 10 V/m at 0.1 cm). These points lead researchers to believe that electric near-fields may play a primary role in neurotransmission and the most evolved brain capabilities.
These findings lead to an intriguing hypothesis at the time of this article. Even accounting for the fact that near-fields do not radiate freely and the propagation losses of brain, skull, and interstitial tissues, they are still difficult to measure at the surface of the skull using current non-invasive technologies. Why is this? The hypothesis is that the structure of nerve fiber volumes is such that it cloaks the continued penetration of the electric near-fields and makes sensing at the surface of the skull difficult. This structure is also possibly what may help to protect the brain from the increasing levels of electromagnetic radiation in the environment.
What makes the hypothesis intriguing is that the near-field is not at all like the far-field, the region associated with typical radio wave propagation and that, contrary to the far-field, the near-field has proved difficult to control and exploit with the current generation of materials. New, synthetic materials called metamaterials, however, are quite a different story10. Our work in this area was invited for a Keynote Presentation at the World Conference & Expo on Biomedical Engineering last July. Main points from the presentation include the following:
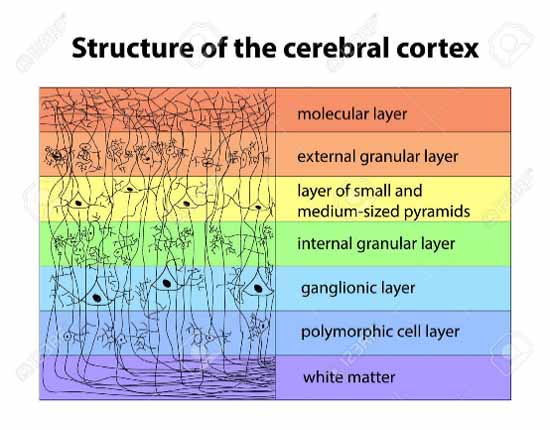
- As an example of brain nerve fiber volume structure, consider the cerebral cortex. The graphic below depicts the many levels of columnar structure that occur on a length scale between approximately the atomic scale and the electric near-field wavelengths:
- Metamaterials are also structured on a length scale between approximately the atomic scale and the wavelength and have entirely novel electric and magnetic properties not believed to be available in nature, such as a negative refractive index.
- Metamaterials are known to interact strongly with near-fields and capture them on neighboring surfaces through short wavelength resonances. This extraordinary property gives a new dimension of control in electromagnetism.
Is the structure of certain brain volumes like that of synthetic metamaterials and responsible for the difficulty in sensing brain near-fields at the surface of the scalp? If so, would these living tissue volumes act like a new class of materials that we might call bio-metamaterials? There has been virtually no research that has been conducted on the electromagnetic properties of human brain tissue structure in the wavelength range [300,2500] nm. In terms of synthetic metamaterials, most of the demonstrators have operated in the GHz region of the spectrum where the engineering is easier. Metamaterial structures exhibiting electromagnetic properties at THz frequencies have been built and at optical frequencies, metamaterial designs exploiting plasma and other resonances are waiting to be exploited. This area of advanced material design is extremely exciting, and we are certain it will provide additional insight into the structure and function of the human brain.
Developing individualized therapy for patients with neurological and neuropsychiatric disorders is the gold standard, a standard that we believe can only be reached by understanding a patient’s electric near-field signatures. This is also the view of other researchers in the field who have adopted rational designs. The “platinum standard” would be to provide individualized therapy in a non-invasive manner with high efficacy and little or no side effects. A futuristic look at this area which is just gaining traction in the realm of clinical medicine is to have minimally invasive devices for diagnosis and treatment that are individually programmed and adaptable to change. Along the path to a non-invasive, clinically accurate medical device is the notable Draper Labs Gemstone11, a 32 channel, scalable, networkable, implantable wireless neurostimulator.
Understanding these extraordinary near-field networks and the possibly bio-metamaterial human brain structure in which they operate will also enable the design of non – von Neumann computing machine architectures and lifelike protheses that benefit from the manner in which nerve fibers interact and form a neurological network. A recent article in Nature Neuroscience asked the question, “What makes the human brain special?” Our work proposes a paradigm shift that leads to the answer, “It is the myelinated nerve fibers of the human brain and their ability to support both intra-fiber salutatory conduction and inter-fiber interaction enabled by electric near-fields.”
References
- S.D. Morgera, “Reactive near field electromagnetic axonal communication channels and their role in neurodegenerative diseases,” in Proc. 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milano, Italy, August 25-29, 2015, pp. 2307-2310. DOI: 10.1109/EMBC.2015.7318854
- S.D. Morgera, S.D., The primary role of the electric near-field in brain function, Electric Field, M.S. Kandelousi, Ed. London: IntechOpen, 2018, pp. 255-282. doi: 10.5772/intechopen.71945.2018.
- A. Chawla, On axon-axon interaction via currents and fields (2017). PhD Dissertation. Tampa: FL: University of South Florida, 2017. (https://scholarcommons.usf.edu/etd/6812).
- A. Frau-Pascual, A. Yendiki, B. Fischl, and I. Aganj, “Global quantification of the structural brain connectivity,” 70th Annual Meeting of the MGH Scientific Advisory Committee Poster Session Abstracts, March 28, 2018.
- A. Frau-Pascual, A. Yendiki, B. Fischl, and I. Aganj, “Global quantification of structural brain connectivity,” in Proc. of the Annual Meeting of the Organization for Human Brain Mapping, Singapore, 2018.
- F. Fröhlich and D.A. McCormick, “Endogenous Electric Fields May Guide Neocortical Network Activity,” Neuron, 67(1), pp. 129-143, July 2010. doi: 10.1016/j.neuron.2010.06.005.
- H. Zhang, D. Salo, D.M. Kim, S. Komarov, Y-C. Tai, and M.Y. Berezin, “Penetration depth of photons in biological tissues from hyperspectral imaging in shortwave infrared in transmission and reflection geometries,” J. Biomed. Opt., 21(12), p. 12606-(1-10), December 2016. doi: 10.1117/1.JBO.21.12.126006.
- D.S. Tuch, V.J. Wedeen, J.S. George, and J.W. Belliveau, “Conductivity tensor mapping of the human brain using diffusion tensor MRI,” in Proc. of the National Academy of Sciences, 98(20), pp. 11697-11701, 2001. DOI: 10.1073/pnas.171473898
- F. Fröhlich, “Endogenous and exogenous electric fields as modifiers of brain activity: rational design of noninvasive brain stimulation with transcranial alternating current stimulation,” Dialogues Clin. Neurosci., 16(1), pp. 93-102, March 2014.
- I. Malkiel, M. Mrejen, A. Nagler, U. Arieli, L. Wolf and H. Suchowski, “Plasmonic nanostructure design and characterization via deep learning,” Light: Science & Applications, 7:60, 2018. DOI: 10.1038/s41377-018-0060-7.
- C.A. Segura, et al., “Gemstone – A Networkable Implantable Wireless Neurostimulator,” iMAPS New England 45th Symposium & Expo., Boxborough, MA, May 1, 2018.
Acknowledgements
The author would like to acknowledge the support of this work by USF and the Center of Excellence for Aging & Brain Repair and Byrd Alzheimer’s Institute and the high technology and medical device companies that have reviewed this work. The accomplishments of Dr. Harry Orf, previously VP at Scripps Florida and now SVP at MGH Research, and Dr. Babak Kateb, Neurosurgeon and CEO and Science Director of the Society for Brain Mapping & Therapeutics (SBMT) served as inspirations for this work. Requests for further information on this work may be directed to Dr. Sal Morgera at sdmorgera@usf.edu.
Biography

Dr. Salvatore Domenic Morgera (https://en.wikipedia.org/wiki/Salvatore_D._Morgera) has focused on networks his entire professional life. He, and the teams he has directed, are responsible for the Canadian oceanographic data gathering networks, the acoustic networks for American submarine tactical and strategic communications, the worldwide CAT3 auto-landing networks for commercial aircraft, and the military wireless networks used in sensitive and challenging parts of the world. In the last decade, he has turned his attention to living networks, the neurological networks of the brain. For more information, see his TEDx talk at USF last September. Dr. Sal Morgera is an IEEE Life Fellow, AAAS Fellow, and IET Fellow. Hi is Professor of Electrical Engineering and Biomedical Engineering at the University of South Florida, Emeritus Professor at Universities in Canada and the US, and Director of the Global Center for Neurological Networks. He received his Bachelor, Master, and Ph.D. degrees from Brown University.


