RESEARCH
Jose L. Contreras-Vidal, Yongtian He, Atilla Kilicarslan, Jose M. Azorín
Trauma to neural systems such as stroke and spinal cord injury (SCI) could irreversibly affect one’s ability to walk. Ambulation and rehabilitation after injuries has long been a research focus with great significance for patients to improve their quality of life. With recent advances in robotic technologies, lower-limb powered exoskeletons have emerged as an assistive and rehabilitative tool for disabled individuals to walk again. The US Food and Drug Administration (FDA) has cleared three exoskeleton devices (ReWalk from ReWalk Robotics, Israel; Indego from Parker Hannifin, USA; and Ekso from Ekso Bionics, USA; see Figures 1D-1F) to market as Class II medical devices in the United States. The interfaces between users and exoskeletons are usually implemented with a combination of buttons, joystick, and sensors to monitor body movement. For instance the ReWalk exoskeleton is shared-controlled by the user shifting body weight alternating between two legs, and also by a wrist-worn device like a digital watch. Brain-machine interfaces (BMIs) on the other hand, control devices directly by interpreting brain activities associated with the user’s motor intent. BMIs make context-based decisions from the users’ cognitive-motor states, thus allowing direct and voluntary operation of the exoskeleton beyond user’s diminished physical, cognitive or sensory capabilities.
BMI technologies have been developed using several different brain recording methods that can be grouped into two categories: invasive and non-invasive. Invasive methods such as intra-cortically implemented electrode arrays have high signal-to-noise ratio (SNR), and have led to proof of concept demonstrations in upper limb BMIs in both non-human primates and people with tetraplegia. So far invasive BMIs have yet to be demonstrated upon people with lower limb disabilities. Invasive approaches face the risk of surgical complications and infections, and the challenge of maintaining long-term stable chronic recordings. To mitigate or eliminate safety risks and reach a broader population, the scalp electroencephalogram (EEG) has been used a non-invasive alternative for BMI applications (Figure 1A). Progress has been made in controlling virtual objects, upper body robotic devices, wheelchairs, etc. Recently, EEG-based BMI control of powered lower-limb exoskeletons has been proposed for the restoration and rehabilitation of gait. This communication focuses on the state-of-the-art and next steps in the development of clinical BMI systems for the control of powered lower-limb exoskeletons.

Figure 1. Some of the notable powered lower-limb exoskeletons. (A) Two researchers wearing exoskeletons (left: H2, right: Rex) and wireless EEG recording devices; (B) Lokomat; (C) a research study that measured EEG of users of NASA X1; (D) Indego; (E) ReWalk; (F) Ekso. Photos B and D-F are provided through courtesy of the respective companies.
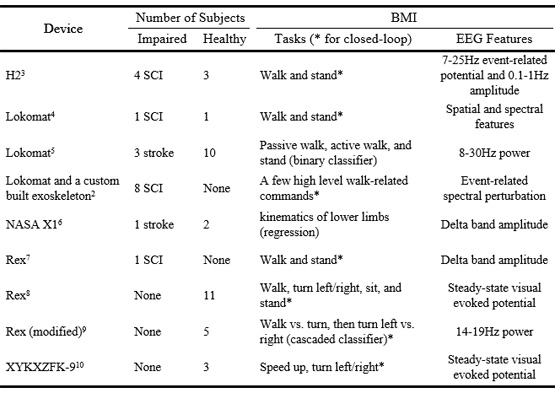
A roadmap for the development of clinical BMIs to lower-limb exoskeletons was proposed in 2013.1 Since then, studies have been published with various devices, BMIs, and protocols. Literature review was conducted in PubMed database with keywords “(EEG OR fNIR) AND (Exoskeleton OR robot OR orthosis) AND (Walk or Lower or Leg or Ambulation)”. One more publications from Europe were manually identified. Table 1 summarizes studies of BMI systems relating to the control of lower-limb exoskeletons.
All nine studies chose EEG as the non-invasive recording method. Six studies used commercial devices (Rex from Rex Bionics, New Zealand, and Lokomat from Hocoma, Switzerland; see Figure 1A right and 1B). Rex is the only device in this category with the ability to self-balance. Lokomat is a rehabilitation device fixed on a treadmill, and thus not an exoskeleton as defined by the FDA. Nonetheless it provides similar functionalities as other devices do, with the exception of ambulation. NASA X1 (US; Figure 1C), H2 (Spain; Figure 1A left), and a custom built device used in Donati et al.’s study2 are three other devices that are still in research phase. XYKXZFK-9 (China) is an inclined platform for users to lie on. It exercises subjects’ legs by moving their feet both vertically and horizontally along the plane.
Six studies included subjects with impaired motor functions including SCI and stroke (18 motor-impaired and 35 healthy subjects in total); seven of them had real time closed-loop control, which is the gold standard for evaluating BMI performance; and only four studies tested their closed-loop BMIs with subjects with disabilities. All closed-loop studies implemented discrete control, allowing their users to choose from a set of preprogrammed robot tasks.
Table 1. State-of-the-art of BMIs for lower-limb exoskeletons
The accuracy and reliability of the decoders are arguably the bottleneck of practical applications of lower-limb BMI systems. Currently most studies have been proof-of-concept. The focus has been on feasibility rather than performance. EEG inevitably has relatively low SNR compared to invasive methods, thus requiring sophisticated artifact removal techniques. Nonetheless, evidence suggests that EEG recordings during walking display distinct features that differ from that in standing, and more importantly, are coupled with the gait cycle.11 Kilicarslan et al. first successfully demonstrated the paradigm of combining BMI with an lower-limb exoskeleton. Moderately high accuracy was achieved in a binary classification between the states of walk and stop.7 Future studies should take advantage of recent progress in walking-related EEG artifact removal methods to improve their performance, and focus on long-term performance in clinical populations.
Exoskeletons have the potential to bring clinical benefits across multiple physiological systems to users with disabilities. Recovery was observed after weeks of training in some clinical studies.12 Moreover, closed-loop BMIs require voluntary effort from the patient to drive the interface, thus ensuring user engagement and promoting cortical plasticity by providing feedback. Studies have reported induced plasticity in upper limb motor functions through BMI protocols.13 Whether BMI-controlled exoskeletons could rehabilitate individuals with walking difficulties remains to be shown. Only one study has measured the clinical benefits of an intervention that included BMI-based exoskeleton training.2 It showed that following 12 months of mixed training that included BMI, virtual reality, tactile feedback, and exoskeletons, all eight SCI patients experienced neurological improvements in somatic sensation. Patients also regained voluntary motor control in key muscles below the SCI level, resulting in marked improvement in their walking index. As a result, 50% of these patients were upgraded to an incomplete paraplegia classification. However, this study mixed various interventions and lacked a control group, making it difficult to assess the extent of contribution of BMI training alone to patients’ recovery.
The risks associated with this new BMI-exoskeleton paradigm are also poorly understood. Injuries resulting from falls are a major safety concern. Moreover, joint misalignment has been reported to cause skin abrasion, sores, hairline fractures and even bone fractures to exoskeleton users. Integrating BMIs with powered exoskeletons increases the complexity of the system, including the need for self-balancing and shared autonomy of these complex human-machine systems. Recent international meetings on wearable robots (e.g. The International Symposium on Wearable Robotics, and Exoskeleton Technical Interchange Meeting) and clinical BMI systems14 have concluded the need for developing standards, interoperability and data sharing mechanisms to speed up the translation of these systems to end users. In addition, research on shared autonomy and automated gait intention detection systems (Association for the Advancement of Artificial Intelligence, 2016 Fall Symposium Series) are critical to the BMI field and long-term deployment of these systems.
Acknowledgments
This work is supported in part by Mission Connect – A TIRR Foundation to J.C.V., and a Fulbright/Spanish Ministry of Education Visiting Scholar Academic Year 2016/2017 (PRX16/00014) to J. A.
References
- Contreras-Vidal, J. L. & Grossman, R. G. NeuroRex: A clinical neural interface roadmap for EEG-based brain machine interfaces to a lower body robotic exoskeleton. Proc. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. EMBS 1579–1582 (2013). doi:10.1109/EMBC.2013.6609816
- Donati, A. R. C. et al. Long-Term Training with a Brain-Machine Interface-Based Gait Protocol Induces Partial Neurological Recovery in Paraplegic Patients. Sci. Rep. 6, 30383 (2016).
- López-Larraz, E. et al. Control of an Ambulatory Exoskeleton with a Brain–Machine Interface for Spinal Cord Injury Gait Rehabilitation. Front. Neurosci. 10, 359 (2016).
- Do, A. H., Wang, P. T., King, C. E., Chun, S. N. & Nenadic, Z. Brain-computer interface controlled robotic gait orthosis. J. Neuroeng. Rehabil. 10, 111 (2013).
- García-Cossio, E. et al. Decoding Sensorimotor Rhythms during Robotic-Assisted Treadmill Walking for Brain Computer Interface (BCI) Applications. PLoS One 10, e0137910 (2015).
- He, Y. et al. An integrated neuro-robotic interface for stroke rehabilitation using the NASA X1 powered lower limb exoskeleton. in 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society 3985–3988 (IEEE, 2014). doi:10.1109/EMBC.2014.6944497
- Kilicarslan, A., Prasad, S., Grossman, R. G. & Contreras-Vidal, J. L. High accuracy decoding of user intentions using EEG to control a lower-body exoskeleton. Conf. Proc. … Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. IEEE Eng. Med. Biol. Soc. Annu. Conf. 2013, 5606–9 (2013).
- Kwak, N.-S., Müller, K.-R. & Lee, S.-W. A lower limb exoskeleton control system based on steady state visual evoked potentials. J. Neural Eng. 12, 56009 (2015).
- Lee, K., Liu, D., Perroud, L., Chavarriaga, R. & Millán, J. del R. A brain-controlled exoskeleton with cascaded event-related desynchronization classifiers. Rob. Auton. Syst. 1–9 (2016). doi:10.1016/j.robot.2016.10.005
- Xin Zhang et al. An EEG-driven Lower Limb Rehabilitation Training System for Active and Passive Co-stimulation. in 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) 4582–4585 (IEEE, 2015). doi:10.1109/EMBC.2015.7319414
- Gwin, J. T., Gramann, K., Makeig, S. & Ferris, D. P. Electrocortical activity is coupled to gait cycle phase during treadmill walking. Neuroimage 54, 1289–96 (2011).
- Contreras-Vidal, J. L. et al. Powered exoskeletons for bipedal locomotion after spinal cord injury. J. Neural Eng. 13, 31001 (2016).
- Ang, K. K. et al. A Randomized Controlled Trial of EEG-Based Motor Imagery Brain-Computer Interface Robotic Rehabilitation for Stroke. Clin. EEG Neurosci. 46, 310–320 (2015).
- Bowsher, K. et al. Brain–computer interface devices for patients with paralysis and amputation: a meeting report. J. Neural Eng. 13, 23001 (2016).
About the Authors
 Jose L. Contreras-Vidal (M’88–SM’08) received the Engineer’s degree from Tecnologico de Monterrey (Mexico, 1987), the M.S.E.E. from the University of Colorado (Boulder, 1990), and the Ph.D. degree from Boston University (Boston, 1994). He was an Assistant and Associate Professor at the University of Maryland, College Park, in 1999 and 2005, respectively. He is Cullen Distinguished Professor of Electrical and Computer Engineering at the University of Houston, Houston, TX. He is Associate Editor for IEEE Human Machine Systems, Neural Networks, Frontiers in Neuroprosthetics, and the International Journal of Mobile Human Computer Interaction and member of the IEEE EMBS and SMC Societies.
Jose L. Contreras-Vidal (M’88–SM’08) received the Engineer’s degree from Tecnologico de Monterrey (Mexico, 1987), the M.S.E.E. from the University of Colorado (Boulder, 1990), and the Ph.D. degree from Boston University (Boston, 1994). He was an Assistant and Associate Professor at the University of Maryland, College Park, in 1999 and 2005, respectively. He is Cullen Distinguished Professor of Electrical and Computer Engineering at the University of Houston, Houston, TX. He is Associate Editor for IEEE Human Machine Systems, Neural Networks, Frontiers in Neuroprosthetics, and the International Journal of Mobile Human Computer Interaction and member of the IEEE EMBS and SMC Societies.
 Yongtian He, IEEE student member, is pursuing his PhD degree in Electrical Engineering in the Non-invasive Brain-machine Interface Systems Laboratory in University of Houston. He received his B.S. in Electronics and Communication Engineering from Huazhong University of Science and Technology (2012, China). He was an intern researcher at Emotiv Inc. (2016, San Francisco). His research topics include EEG signal processing, brain-machine interfaces, human gait rehabilitation, and regulatory science for medical exoskeletons.
Yongtian He, IEEE student member, is pursuing his PhD degree in Electrical Engineering in the Non-invasive Brain-machine Interface Systems Laboratory in University of Houston. He received his B.S. in Electronics and Communication Engineering from Huazhong University of Science and Technology (2012, China). He was an intern researcher at Emotiv Inc. (2016, San Francisco). His research topics include EEG signal processing, brain-machine interfaces, human gait rehabilitation, and regulatory science for medical exoskeletons.
 Atilla Kilicarslan, IEEE member, is a Research Assistant Professor at the University of Houston, Electrical and Computer Engineering Department. He received his M.Sc. from the Technical University of Istanbul on System Dynamics and Control program and worked on biomimetic robotic systems, and Ph.D. from the University of Houston, Mechanical Engineering Department working on advanced control applications to smart materials and medical robotics. His current research focus is on the algorithmic formulation and real-time implementation of Brain Machine Interface systems to robotic exoskeletons, including methods for real-time EEG artifact cleaning.
Atilla Kilicarslan, IEEE member, is a Research Assistant Professor at the University of Houston, Electrical and Computer Engineering Department. He received his M.Sc. from the Technical University of Istanbul on System Dynamics and Control program and worked on biomimetic robotic systems, and Ph.D. from the University of Houston, Mechanical Engineering Department working on advanced control applications to smart materials and medical robotics. His current research focus is on the algorithmic formulation and real-time implementation of Brain Machine Interface systems to robotic exoskeletons, including methods for real-time EEG artifact cleaning.
 José M. Azorín (SM’16) is Director of the Brain-Machine Interface Systems Lab and Associate Professor of the Systems Engineering and Automation Department at Miguel Hernández University of Elche (Spain). He holds an M.Sc from the University of Alicante (1997, Spain) and a Ph.D. from the Miguel Hernández University of Elche (2003, Spain). His current research interests are Brain-Computer Interfaces, Neuro-robotics, Assistive Robotics and Rehabilitation Robotics. He is a member of the publications board of the IEEE Robotics and Automation Society, advisory committee of the IEEE Systems Council, Distinguished Lecturer of the IEEE Systems Council, and IEEE EMBS and SMC Societies.
José M. Azorín (SM’16) is Director of the Brain-Machine Interface Systems Lab and Associate Professor of the Systems Engineering and Automation Department at Miguel Hernández University of Elche (Spain). He holds an M.Sc from the University of Alicante (1997, Spain) and a Ph.D. from the Miguel Hernández University of Elche (2003, Spain). His current research interests are Brain-Computer Interfaces, Neuro-robotics, Assistive Robotics and Rehabilitation Robotics. He is a member of the publications board of the IEEE Robotics and Automation Society, advisory committee of the IEEE Systems Council, Distinguished Lecturer of the IEEE Systems Council, and IEEE EMBS and SMC Societies.


